- Blog
- About
- Contact
- Premiere Pro 14.2
- Ookla Speed Test Server
- Photoshop Tnt
- Fairy Tail Dragon Cry English Sub
- Joomla Reservation
- Burp Portswigger
- Plusnet Broadband Speed Checker
- Morgana
- League Of Legends Release
- Burp Suite Http Request Smuggling
- Burp Suite Wordpress
- Carbon Protons
- Burp Suite Facebook
- Traktor Ableton
- Tufts Outlook
- Prosoniq Orange Vocoder Torrent
- Adobe Photoshop Cc 2020 Full
- Adobe Mixamo
- Lol Wild
- Spd Service Tool V1.0.0 Free Download
- Wild Rift Upcoming Champions
- How To Change Lenovo Laptop Serial Number In Bios
- Rar 7
- Mybroadband Speed Test
- Fusion 360 Grbl
- Speed Test Internet Online Free
- Dockmate Twist
- Burp Suite License Key Free
- Google Fiber Test Speed
- Champion Select
- Photoshop 2020 Google Drive
- Legends Of Runeterra Ranked
- Vst Ableton 10
- Rar English
- Download Bajaj Boxer Service Manual Software
- Burp Suite Send Multiple Requests
- Photoshop Classic
- Dsl Speed Test Ookla
- Ableton Live 10 Edu
- Rar File Opener Online
- Ableton Live Ipad
- Rising Action
- Red Alert 2 Windows 10 Black Screen
- Furyhorn Veigar
- Speedtest Cablevision
The nucleus of carbon-12 contains six protons and six neutrons, resulting in an atomic mass of 12 amu.: The nucleus of carbon-13 contains six protons and seven neutrons, resulting in an atomic mass of 13 amu.: The nucleus of carbon-14 contains six protons and eight neutrons, resulting in.
Carbon is one of the chemical elements. Along with hydrogen, nitrogen, oxygen, phosphorus, and sulfur, carbon is a building block of biochemical molecules ranging from fats, proteins, and carbohydrates to active substances such as hormones. All carbon atoms have a nucleus containing six protons. Ninety-nine percent of these also contain six neutrons. The 6 proton + 6 neutron atoms are said to have a mass of 12 and are referred to as 'carbon-12.' The nuclei of the remaining one percent of carbon atoms contain not six but either seven or eight neutrons in addition to the standard six protons. They have masses of 13 and 14 respectively and are referred to as 'carbon-13' and 'carbon-14.'
If two atoms have equal numbers of protons but differing numbers of neutrons, one is said to be an 'isotope' of the other. Carbon-13 and carbon-14 are thus isotopes of carbon-12. Isotopes participate in the same chemical reactions but often at differing rates. When isotopes are to be designated specifically, the chemical symbol is expanded to identify the mass (for example, 13C).
- Carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure.
- The element carbon (C) has an atomic number of 6, which means that all neutral carbon atoms contain 6 protons and 6 electrons. In a typical sample of carbon-containing material, 98.89% of the carbon atoms also contain 6 neutrons, so each has a mass number of 12.
(Illustration by Jayne Doucette, Woods Hole Oceanographic Institution)
See real world download speeds for Google Fiber based on over 21.8K speed tests from verified users over the past 12 months. See everything from download speed, to. Google Fiber speed test is performed by performing various speed tests using your Google Fiber internet connection. This happens when our speed test service sends a chunks of bytes to Google Fiber's nearest server. Firstly, we perform a download test in which chunks of bytes are downloaded from remote server to your computer and the transfer. What speeds should I expect? Learn more About. Blog; Careers; Privacy & Terms; Social Media. Google+; Facebook; Twitter; YouTube. Google fiber speed test by ookla.
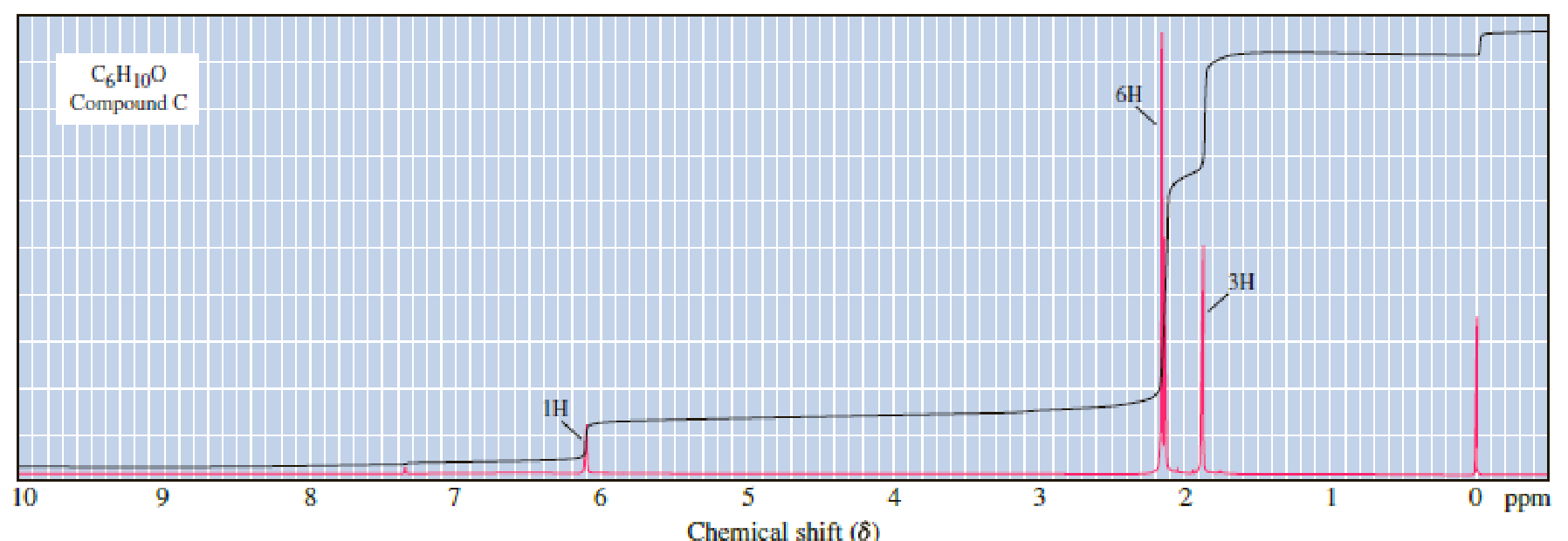
Carbon Protons
Recommended
- Radiocarbon Web-info
Web-info Radiocarbon from University of Waikato Radiocarbon Dating Laboratory, New Zealand - A.E. Lalonde AMS Laboratory at the University of Ottowa
The Canadian Centre for Accelerator Mass Spectrometry at the University of Ottowa. - NOVA Interactive Radiocarbon
An interactive introduction to radiocarbon dating via AMS at NOSAMS. - How does Radiocarbon work?
Scientific American Editor Michael Moyer explains the process of radiocarbon dating.
Both 13C and 14C are present in nature. The former accounts for about 1% of all carbon. The abundance of 14C varies from 0.0000000001% (one part per trillion, a small, but measurable, level) down to zero. The highest abundances of 14C are found in atmospheric carbon dioxide and in products made from atmospheric carbon dioxide (for example, plants). Unlike 12C and 13C, 14C is not stable. As a result it is always undergoing natural radioactive decay while the abundances of the other isotopes are unchanged. Carbon-14 is most abundant in atmospheric carbon dioxide because it is constantly being produced by collisions between nitrogen atoms and cosmic rays at the upper limits of the atmosphere.
The rate at which 14C decays is absolutely constant. Given any set of 14C atoms, half of them will decay in 5700 years. Since this rate is slow relative to the movement of carbon through food chains (from plants to animals to bacteria) all carbon in biomass at earth's surface contains atmospheric levels of 14C. However, as soon as any carbon drops out of the cycle of biological processes - for example, through burial in mud or soil - the abundance of 14C begins to decline. After 5700 years only half remains. After another 5700 years only a quarter remains. This process, which continues until no 14C remains, is the basis of carbon dating.
A sample in which 14C is no longer detectable is said to be 'radiocarbon dead.' Fossil fuels provide a common example. They are derived from biomass that initially contained atmospheric levels of 14C. But the transformation of sedimentary organic debris into oil or woody plants into coal is so slow that even the youngest deposits are radiocarbon dead.
The abundance of 14C in an organic molecule thus provides information about the source of its carbon. If 14C is present at atmospheric levels, the molecule must derive from a recent plant product. The pathway from the plant to the molecule may have been indirect or lengthy, involving multiple physical, chemical, and biological processes. Levels of 14C are affected significantly only by the passage of time. If a molecule contains no detectable 14C it must derive from a petrochemical feedstock or from some other ancient source. Intermediate levels of 14C can represent either mixtures of modern and dead carbon or carbon that was fixed from the atmosphere less than 50,000 years ago.
Signals of this kind are often used by chemists studying natural environments. A hydrocarbon found in beach sediments, for example, might derive from an oil spill or from waxes produced by plants. If isotopic analyses show that the hydrocarbon contains 14C at atmospheric levels, it's from a plant. If it contains no 14C, it's from an oil spill. If it contains some intermediate level, it's from a mixture of both sources.
- The fusion reaction
- Methods of achieving fusion energy
Please select which sections you would like to print:
While every effort has been made to follow citation style rules, there may be some discrepancies. Please refer to the appropriate style manual or other sources if you have any questions.
Our editors will review what you’ve submitted and determine whether to revise the article.
Join Britannica's Publishing Partner Program and our community of experts to gain a global audience for your work! Robert W. ConnProfessor of Applied Science, University of California, San Diego. Founder and principal editor (1986-2001), Fusion Engineering and Design.
Alternative Titles: atomic fusion, fusion, thermonuclear fusion
Carbon Atomic Mass
Nuclear fusion, process by which nuclear reactions between light elements form heavier elements (up to iron). In cases where the interacting nuclei belong to elements with low atomic numbers (e.g., hydrogen [atomic number 1] or its isotopes deuterium and tritium), substantial amounts of energy are released. The vast energy potential of nuclear fusion was first exploited in thermonuclear weapons, or hydrogen bombs, which were developed in the decade immediately following World War II. For a detailed history of this development, seenuclear weapon. Meanwhile, the potential peaceful applications of nuclear fusion, especially in view of the essentially limitless supply of fusion fuel on Earth, have encouraged an immense effort to harness this process for the production of power. For more detailed information on this effort, seefusion reactor.
This article focuses on the physics of the fusion reaction and on the principles of achieving sustained energy-producing fusion reactions.
The fusion reaction
Fusion reactions constitute the fundamental energy source of stars, including the Sun. The evolution of stars can be viewed as a passage through various stages as thermonuclear reactions and nucleosynthesis cause compositional changes over long time spans. Hydrogen (H) “burning” initiates the fusion energy source of stars and leads to the formation of helium (He). Generation of fusion energy for practical use also relies on fusion reactions between the lightest elements that burn to form helium. In fact, the heavy isotopes of hydrogen—deuterium (D) and tritium (T)—react more efficiently with each other, and, when they do undergo fusion, they yield more energy per reaction than do two hydrogen nuclei. (The hydrogen nucleus consists of a single proton. The deuterium nucleus has one proton and one neutron, while tritium has one proton and two neutrons.)
Fusion reactions between light elements, like fission reactions that split heavy elements, release energy because of a key feature of nuclear matter called the binding energy, which can be released through fusion or fission. The binding energy of the nucleus is a measure of the efficiency with which its constituent nucleons are bound together. Take, for example, an element with Z protons and N neutrons in its nucleus. The element’s atomic weightA is Z + N, and its atomic number is Z. The binding energy B is the energy associated with the mass difference between the Z protons and N neutrons considered separately and the nucleons bound together (Z + N) in a nucleus of mass M. The formula is B = (Zmp + Nmn − M)c2, where mp and mn are the proton and neutron masses and c is the speed of light. It has been determined experimentally that the binding energy per nucleon is a maximum of about 1.4 10−12joule at an atomic mass number of approximately 60—that is, approximately the atomic mass number of iron. Accordingly, the fusion of elements lighter than iron or the splitting of heavier ones generally leads to a net release of energy.
Get a Britannica Premium subscription and gain access to exclusive content. Subscribe Now
Two types of fusion reactions
Fusion reactions are of two basic types: (1) those that preserve the number of protons and neutrons and (2) those that involve a conversion between protons and neutrons. Reactions of the first type are most important for practical fusion energy production, whereas those of the second type are crucial to the initiation of star burning. An arbitrary element is indicated by the notation AZX, where Z is the charge of the nucleus and A is the atomic weight. An important fusion reaction for practical energy generation is that between deuterium and tritium (the D-T fusion reaction). It produces helium (He) and a neutron (n) and is written D + T → He + n.
Carbon Protons And Neutrons
To the left of the arrow (before the reaction) there are two protons and three neutrons. The same is true on the right.
The other reaction, that which initiates star burning, involves the fusion of two hydrogen nuclei to form deuterium (the H-H fusion reaction):H + H → D + β + + ν, where β + represents a positron and ν stands for a neutrino. Before the reaction there are two hydrogen nuclei (that is, two protons). Afterward there are one proton and one neutron (bound together as the nucleus of deuterium) plus a positron and a neutrino (produced as a consequence of the conversion of one proton to a neutron).
Both of these fusion reactions are exoergic and so yield energy. The German-born physicist Hans Bethe proposed in the 1930s that the H-H fusion reaction could occur with a net release of energy and provide, along with subsequent reactions, the fundamental energy source sustaining the stars. However, practical energy generation requires the D-T reaction for two reasons: first, the rate of reactions between deuterium and tritium is much higher than that between protons; second, the net energy release from the D-T reaction is 40 times greater than that from the H-H reaction.
Quick Facts
- key people
- related topics
